
IT’s ABOUT TIME WE DISCUSS SAFETY
A young woman, Anna Crail, was flying on 19 March of this year on an Alaska Airlines flight from Seattle to Honolulu. About 90 minutes prior to landing, her iPhone 6 suddenly broke out in flames causing panic in mid flight. The fire was rapidly extinguished by the...
A young woman, Anna Crail, was flying on 19 March of this year on an Alaska Airlines flight from Seattle to Honolulu. About 90 minutes prior to landing, her iPhone 6 suddenly broke out in flames causing panic in mid flight. The fire was rapidly extinguished by the flight attendants, but not without leaving the airline and the FAA searching for answers. This is one of several safety-related battery incidents that are becoming increasingly common. There are countless reports on hoverboards that are catching fire. While safety-related incidents involving Apple iPhones appear to be sparse in the media, there are increasing reports of Android-type mobile devices posing serious safety hazards, especially in Asian geographies. This post provides first insights on the factors that impact the safety of the lithium-ion battery in mobile devices.
First, let’s start with some background material. Three key design factors heavily influence the lithium-ion battery in a smartphone: i) Higher charge capacity, i.e., more mAh; ii) Faster charging ; and lastly iii) thin profiles.
Higher capacity is driving an increase in energy density at the rate of about 5 to 7% per year. The energy density of lithium-ion batteries when the first iPhone was launched in 2007 was near 400 Wh/l. Today’s state-of-the-art mobile batteries are in the range of 600 to 700 Wh/l. These higher energy densities are associated with two important physical parameters: higher terminal voltage (near 4.4V, up from 4.2V ten years ago) and significantly higher current flux inside the batteries (a lot more ions are making the journey between the electrodes) elevating the risk of damage within the battery.
Fast charging is now an expected feature in mobile devices, at least those on the higher end of the spectrum. A charge rate of 1C corresponds to a 50% charge in 30 minutes…1C or faster is becoming the norm. Faster charging also means a lot more ions are making the journey within the battery between the two opposite electrodes. Again, faster charging = higher risk of battery damage.
Lastly, thin profiles of smartphones are pushing the battery to ever thinner dimensions. The next generation of smartphones are employing batteries that are a mere 3 to 4 mm thick, with this figure being pushed down even further where possible. Thin batteries create a slew of headaches for engineers…their performance tends to be inconsistent; manufacturing non-uniformities are amplified; and the current flux (and corresponding ion density) within the battery is also pushed to higher levels.
So what do these really mean? they mean that the perfect safety storm is brewing if proper care, battery intelligence, and diagnostics are not implemented. One of the first consequences of higher energy density (especially higher operating voltages near 4.4V) is the increased risk of formation of lithium metal on the carbon anode (the negative electrode in the battery during charging). This is called lithium plating. Combine high energy density with fast charging and thin batteries, and the risk of lithium plating becomes dangerously significant. But lithium metal (i.e, in its molecular form, not in its ionic form) is highly flammable especially in the presence of oxidants. Additionally, spurs of lithium metal can cause electrical shorts within the battery…both of these mechanisms have seriously hazardous consequences.
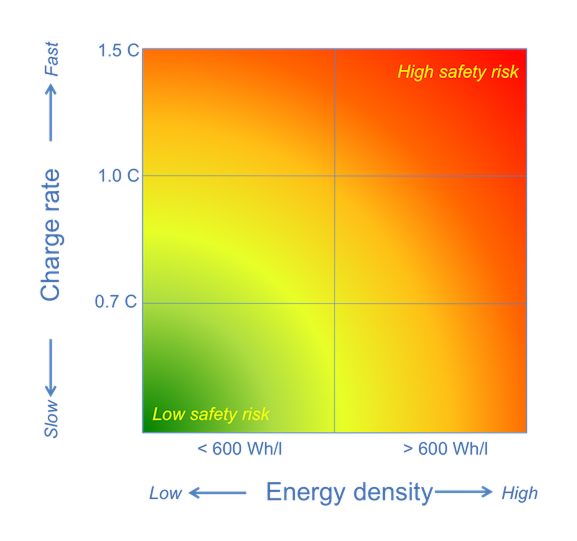
The next figure illustrates the safety risk and its relationship to charge rate and energy density. Right around 1C and 600 Wh/l, the battery may become a safety hazard, especially in the absence of proper and diligent designs. Some battery manufacturers are better than others, with batteries made by Chinese manufacturers being the most prone to increased safety risks. Some device manufacturers (OEM) choose to sacrifice some battery specifications to gain a little safety margin. For example, some OEMs reduce the operating voltage of the battery as it ages, for example, from 4.4V down to 4.35V or less. This means that you will be robbed of mAh without being told. Your smartphone may have a great battery (say for example, 3000 mAh) at the beginning of its life (and when it operated at 4.4V), but a few months in its operation, the maximum voltage is intentionally reduced to 4.35V thereby reducing the capacity by 150 – 200 mAh; in other words, your battery is now only about 2800 mAh. Ouch! That’s not good, especially when you are not aware of it.
The safety risk also depends on temperature, rising rapidly with lower temperatures…and by low temperatures, I really don’t mean sub-freezing temperatures. The vast majority of battery safety tests are conducted at room temperature, usually near 25 °C (77 °F). What is considered as “low temperature” for a battery is 10 °C (50 °F) or lower. Right around this temperature range, the probability of plating of lithium metal soars creating serious hazards.
For the time being, batteries catching fire have been mostly limited in frequency and consequences. But with rising energy density and charge rates, the safety hazard is slated to become a lot more serious in the near future. Look for smartphone OEMs that are investing in the proper solutions to give you an excellent battery experience AND a safe one too.
